Worldwide H1N1 (Swine Flu) Infection Data
Infection and Death Rate Per Million of Population for many countries
U.S. Lab-Verified Swine Flu Infections for each US State
World Lab-Verified Swine Flu Infections
Death Rate per Infection
Some selected "Death Rate per Infection" (copied Oct 29, 2009)
Country Cases Deaths Frequency Solomon Islands 3 1 1 in 3 Brazil 30206 2105 1 in 14 United States 70034 1170 1 in 60 China (Hong Kong) 31211 36 1 in 867 China (Mainland) 35664 3 1 in 11,888
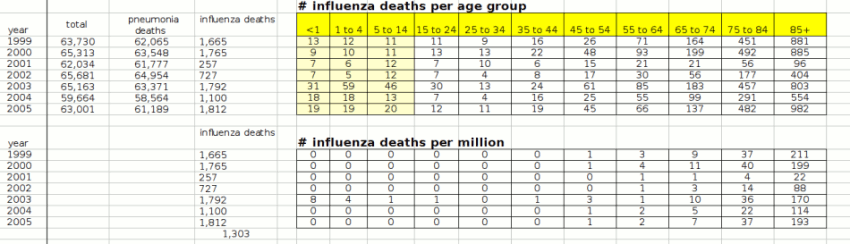
Deaths from Flu in previous years
http://thegranddisillusion.files.wordpress.com/2009/10/monthlyswinefludeaths.jpg?w=578&h=302
https://www.medicalcountermeasures.gov/BARDA/MCM/panflu/factsheet.aspx
HHS Contracting Activities
Since 2004, HHS has contracted with manufacturers that currently hold U.S. licenses for flu vaccine as part of the National Strategy for Pandemic Influenza. In May 2009, HHS issued new orders on these contracts to produce a bulk supply of vaccine antigen and adjuvant and to produce pilot (also called investigational) lots of a 2009 H1N1 vaccine. Most will be stored in bulk, and a small amount will be prepared as vaccine for use in clinical studies to evaluate vaccine safety and the dosage required for a protective effect. This research will include studies with adjuvant to determine its safety and the effect it would have on the immune system’s response.
Orders for Bulk Supply of H1N1 Influenza Vaccine Antigen and Adjuvant: May 22, 2009
|
Manufacturer |
Bulk Vaccine Antigen |
Oil-In-Water Bulk Adjuvant |
|
Novartis |
$150 million |
$139 million |
|
GlaxoSmithKline |
$ 38 million |
$144 million |
|
Sanofi Pasteur |
$191 million |
|
|
CSL Biotherapies |
$180 million |
|
|
MedImmune |
$ 90 million |
|
|
Total |
$649 million |
$283 million |
Orders for Bulk Supply of H1N1 Influenza Vaccine Antigen and Adjuvant: July 9, 2009
|
Manufacturer |
Bulk Vaccine Antigen |
Bulk Virus Concentrate/FFF |
Oil-In-Water Bulk Adjuvant |
|
Sanofi Pasteur |
$61,425,000 |
0 |
0 |
|
GSK |
$0 |
0 |
$71,400,000 |
|
Novartis |
$346,334,450 |
0 |
$343,810,470 |
|
CSL |
$0 |
0 |
0 |
|
MedImmune |
$0 |
$61,008,000 |
0 |
|
Total |
$407,759,450 |
$61,008,000 |
$415,210,470 |
CBS News Exclusive: Study Of State Results Finds H1N1 Not As Prevalent As Feared
Source: http://www.cbsnews.com/stories/2009/10/21/cbsnews_investigates/main5404829.shtml
Oct. 21, 2009
(CBS) If you've been diagnosed "probable" or "presumed" 2009 H1N1 or "swine flu" in recent months, you may be surprised to know this: odds are you didn’t have H1N1 flu.
In fact, you probably didn’t have flu at all. That's according to state-by-state test results obtained in a three-month-long CBS News investigation.
The ramifications of this finding are important. According to the Centers for Disease Control and Prevention (CDC) and Britain's National Health Service, once you have H1N1 flu, you're immune from future outbreaks of the same virus. Those who think they've had H1N1 flu -- but haven't -- might mistakenly presume they're immune. As a result, they might skip taking a vaccine that could help them, and expose themselves to others with H1N1 flu under the mistaken belief they won't catch it. Parents might not keep sick children home from school, mistakenly believing they've already had H1N1 flu.
Why the uncertainty about who has and who hasn't had H1N1 flu?
Attkisson Blogs: Freedom of Information Stalled at CDC
CBSNews.com report on H1N1
In late July, the CDC abruptly advised states to stop testing for H1N1 flu, and stopped counting individual cases. The rationale given for the CDC guidance to forego testing and tracking individual cases was: why waste resources testing for H1N1 flu when the government has already confirmed there's an epidemic?
Some public health officials privately disagreed with the decision to stop testing and counting, telling CBS News that continued tracking of this new and possibly changing virus was important because H1N1 has a different epidemiology, affects younger people more than seasonal flu and has been shown to have a higher case fatality rate than other flu virus strains.
CBS News learned that the decision to stop counting H1N1 flu cases was made so hastily that states weren't given the opportunity to provide input. Instead, on July 24, the Council for State and Territorial Epidemiologists, CSTE, issued the following notice to state public health officials on behalf of the CDC:
"Attached are the Q&As that will be posted on the CDC website tomorrow explaining why CDC is no longer reporting case counts for novel H1N1. CDC would have liked to have run these by you for input but unfortunately there was not enough time before these needed to be posted (emphasis added)."
When CDC did not provide us with the material, we filed a Freedom of Information request with the Department of Health and Human Services (HHS). More than two months later, the request has not been fulfilled. We also asked CDC for state-by-state test results prior to halting of testing and tracking, but CDC was again, initially, unresponsive.
CBS News Videos Online
Video above: A CBS News producer asks the director of the CDC, Dr. Thomas Frieden, for this information at a press conference on Sept. 19.
While we waited for CDC to provide the data, which it eventually did, we asked all 50 states for their statistics on state lab-confirmed H1N1 prior to the halt of individual testing and counting in July. The results reveal a pattern that surprised a number of health care professionals we consulted. The vast majority of cases were negative for H1N1 as well as seasonal flu, despite the fact that many states were specifically testing patients deemed to be most likely to have H1N1 flu, based on symptoms and risk factors, such as travel to Mexico.
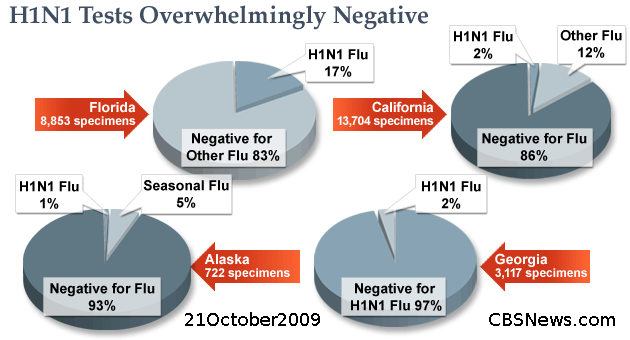
(CBS)
It’s unknown what patients who tested negative for flu were actually afflicted with since the illness was not otherwise determined. Health experts say it’s assumed the patients had some sort of cold or upper respiratory infection that is just not influenza.
With most cases diagnosed solely on symptoms and risk factors, the H1N1 flu epidemic may seem worse than it is. For example, on Sept. 22, this alarming headline came from Georgetown University in Washington D.C.: "H1N1 Flu Infects Over 250 Georgetown Students."
H1N1 flu can be deadly and an outbreak of 250 students would be an especially troubling cluster. However, the number of sick students came not from lab-confirmed tests but from "estimates" made by counting "students who went to the Student Health Center with flu symptoms, students who called the H1N1 hotline or the Health Center's doctor-on-call, and students who went to the hospital's emergency room."
Without lab testing, it's impossible to know how many of the students actually had H1N1 flu. But the statistical trend indicates it was likely much fewer than 250.
CDC continues to monitor flu in general and H1N1 through "sentinels," which basically act as spot-checks to detect trends around the nation. But at least one state, California, has found value in tracking H1N1 flu in greater detail.
"What we are doing is much more detailed and expensive than what CDC wants," said Dr. Bela Matyas, California's Acting Chief of Emergency Preparedness and Response. "We're gathering data better to answer how severe is the illness. With CDC's fallback position, there are so many uncertainties with who's being counted, it's hard to know how much we're seeing is due to H1N1 flu rather than a mix of influenza diseases generally. We can tell that apart but they can't."
After our conversation with Dr. Matyas, public affairs officials with the California Department of Public Health emphasized to CBS News that they support CDC policy to stop counting individual cases, maintaining that the state has the resources to gather more specific testing data than the CDC.
Because of the uncertainties, the CDC advises even those who were told they had H1N1 to get vaccinated unless they had lab confirmation. "Persons who are uncertain about how they were diagnosed should get the 2009 H1N1 vaccine."
That's unwelcome news for a Marietta, Georgia mom whose two children were diagnosed with "probable" H1N1 flu over the summer. She hoped that would mean they wouldn't need the hastily developed H1N1 flu vaccine. However, since their cases were never confirmed with lab tests, the CDC advises they get the vaccine. "I wish they had tested and that I knew for sure whether they had it. I'm not anxious to give them an experimental vaccine if they don't need it."
Speaking to CBS' "60 Minutes," CDC Director Dr. Frieden said he has confidence that the vaccine will be safe and effective: "We're confident it will be effective we have every reason to believe that it will be safe."
However, the CDC recommendation for those who had "probable" or "presumed" H1N1 flu to go ahead and get vaccinated anyway means the relatively small proportion of those who actually did have H1N1 flu will be getting the vaccine unnecessarily. This exposes them to rare but significant side effects, such as paralysis from Guillain-Barre syndrome.
It also uses up vaccine, which is said to be in short supply. The CDC was hoping to have shipped 40 million doses by the end of October, but only about 30 million doses will be available this month.
The CDC did not response to questions from CBS News for this report.
Washington Unplugged: H1N1 Cases Exaggerated?
H1N1 Misdiagnoses Could Have Consequences